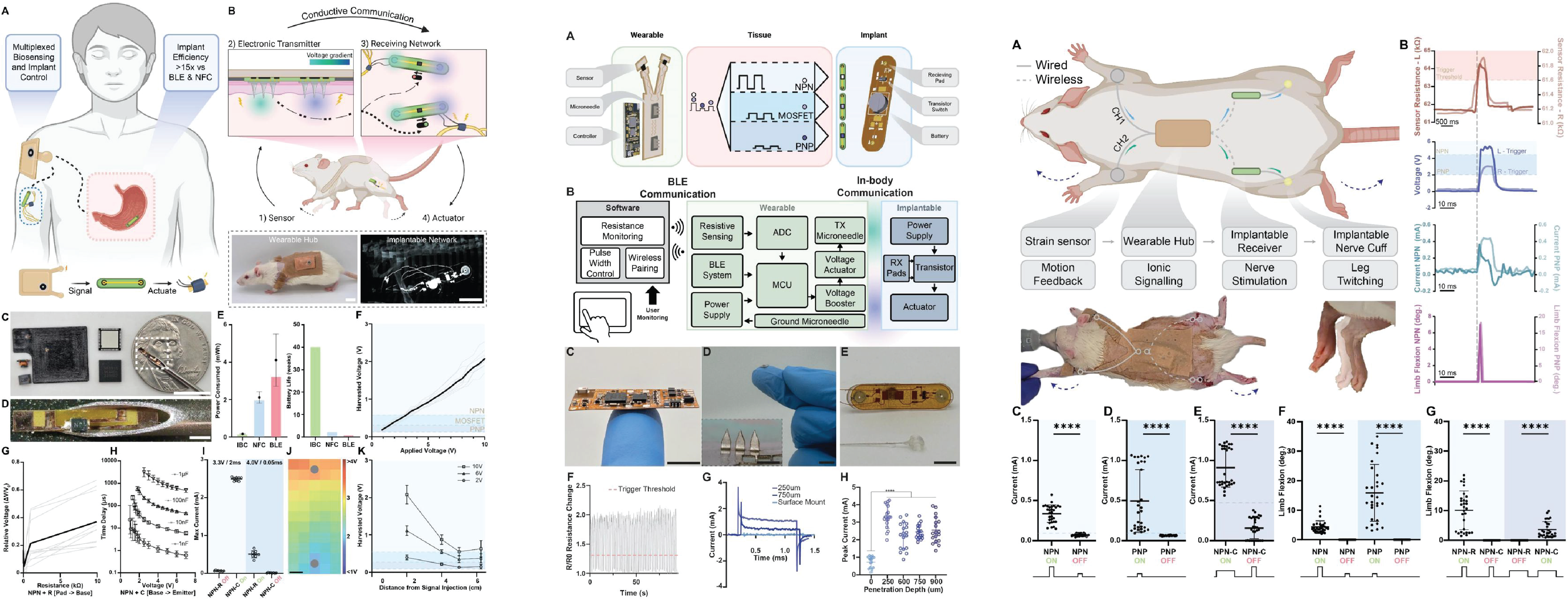
Abstract— Bioelectronics have transformed our capacity to monitor and treat diseases; however, a lack of micrometer-scale, energy efficient communication options limit these devices from forming integrated networks that enable full-body, sensor driven, physiological control. Inspired by our nervous system’s ability to transmit information via ionic conduction, we engineered a Smart Wireless Artificial Nervous System (SWANS) that utilizes the body’s own tissue to transmit signals between wearables and implantables. When SWANS emits signals, it generates voltage gradients throughout the body that selectively turn on implanted transistor switches when exceeding their gate threshold voltages. SWANS’ implantable communication components maintain syringe-injectable footprints and >15x greater power efficiencies than Bluetooth and Near Field Communication. In vivo studies in rats demonstrate SWANS’ ability to wirelessly regulate dual hind leg motor control by connecting electronic-skin sensors to implantable neural interfaces via ionic signaling as well as coordinate bioelectronics throughout the epidermal, subcutaneous, intraperitoneal, and gastrointestinal spaces.
Introduction— Wearable (1, 2), implantable (3–6), and ingestible (7, 8) bioelectronics monitor and treat the world’s most prevalent illnesses including diabetes (9, 10), cancer (11, 12), and heart disease (13, 14); however, almost all bioelectronics work independently from one another, limiting the capacity for biosensors to interact and remotely trigger therapeutics. In-body networks of electronic therapeutics are inherently limited by the large power and size requirements necessary for communication that limit implant battery lifetimes to weeks and necessitate invasive surgical implantation (15–17). One example of sensors and therapeutics working side-by-side is the wearable continuous glucose monitor and insulin pump; yet insulin pumps’ batteries, measuring 8 cm3, only last 7 days partially due to constant Bluetooth Low Energy (BLE) connectivity (18). Other wireless communication and power transfer methods, such as near-field communication (NFC), require bulky external antennae that limit patient adherence and require centimeter-scale alignment with targeted devices (19, 20). Here, we harness the conductive nature of body tissue to transmit communication signals ionically between wearable sensors and implantable devices. In the process, we create an artificial nervous system that: (1) reduces communication components to an emitted square-wave pulse and a single micrometer-scale transistor per implanted device; (2) independently triggers multiple devices throughout the tissue from a multiplexed wearable hub; and (3) extends the battery life of implanted devices compared to NFC and Bluetooth by 15x and 30x, respectively.
Here, we introduce SWANS, the Smart Wearable Artificial Nervous System. SWANS unlocks the potential for long-term closed-loop networks of wearable and in-body therapeutics by providing a framework for communication between dispersed bioelectronics via intertissue ionic conduction (Fig. 1A). To enable effective communication, SWANS’ wearable biosensor hub emits electrical pulses that produce electric fields inside the body; then, the implantable receiving network detects the generated voltage gradient through tissue-interfacing pads to turn on a transistor switch which briefly enables a therapeutic action (Fig. 1B). Each implant possesses a unique transistor circuit that only responds to its corresponding signal. The U.S. Food and Drug Administration has already confirmed the safety and efficacy of in-body ionic communication in humans for other uses (21); for example, Abilify MyCite pills utilize ionic conduction to transmit ingestion event data from an oral drug tablet to a wearable device by recording the presence or absence of an electrical signal in the body (22, 23). Preclinical studies have also utilized ionic conductance to similarly transmit data across tissues from implantable (24), wearable (25), and ingestible (26) sensors. Additionally, engineers have previously attempted to create body-networks of wearable sensors without including implants (27, 28); however, a fully functional body-therapeutic-network requires integration of both sensors and actuators across all tissue depths to enable closed-loop therapies. We demonstrate in vivo in rats that SWANS can wirelessly trigger subcutaneously implanted, intraperitoneally implanted, and gastrointestinally localized actuators via sensors and electronics placed on the epidermis. Furthermore, we demonstrate in vivo proof-of-concept by utilizing the SWANS architecture to wirelessly trigger implantable sciatic nerve stimulators for dual hind-leg animation. By detecting front limb motion and activating hind leg motion, we show that SWANS mimics how nervous systems selectively trigger and transmit targeted signals from one end of the body to another.
 |
Aristide Gumyusenge
|
